This article provides a comprehensive introduction to chloroform as an organic substance, and partners who wish to learn about chloroform can read this article to gain a comprehensive understanding of this substance.
Contents I, Chloroform meaning II, CHCl3 Physical and Chemical Properties III, Special properties of chloroform IV, Chloroform uses 4.1 Organic solvent
4.2 Organic synthesis
4.3 Extraction
4.4 Chemical Analysis 4.5 Deuterium Reagent (Chloroform d)
4.6 Chloracarbon
4.7 Halogenation
4.8 Reduce Sulphuric Acid Carbonation
V, Eight technical routes for the production of chloroform.
I, Chloroform meaning
The chemical formula of Chloroform is CHCl3, which is trichloromethane.
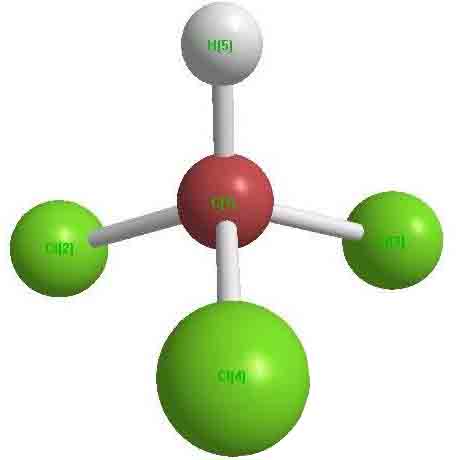 |
Chloroform is trichloromethane. Its molecule has one carbon atom, three chlorine atoms and one hydrogen atom.The structure is a non-orthotetrahedral. |
II, CHCl3 Physical and Chemical Properties
| Relative density |
1.4840 |
Boiling point |
61~62°C |
| Freezing point |
-63.5℃ |
Refractive index |
1.4476 |
| Toxicity |
low, LD50 (rat, oral) 1194mg/kg. Narcotic.Carcinogenic. |
Polarity |
Chloroform is a polar molecule, but the polarity is very weak. |
| Chloroform molar mass |
84.93 g/mol |
Solubility |
Insoluble in water. Soluble in ethanol, benzene, ether, petroleum ether, carbon tetrachloride, carbon disulfide, and oil, etc. |
Appearance
|
Colorless transparent heavy liquid |
Critical temperature |
263.4℃ |
Saturated vapor pressure
|
13.33kPa (10.4℃) |
Critical pressure |
5.47MPa |
III, Special properties of chloroform
Chloroform is sensitive to light and can be oxidized in the presence of light and air to produce the highly toxic phosgene.
Therefore, chloroform should be kept in brown bottles and filled to the bottle to be sealed to avoid exposure to light and air contact.
For medicinal purposes, 1% ethanol should be added to the chloroform to destroy any phosgene that may be generated. |
 |
Toxicity:
Chloroform has strong anesthesia. Because it has toxicity to the heart and liver, clinical has stopped using it as an anesthetic.
What does chloroform smell like?
Chloroform smells similar to ether, the smell is a peculiar odor.
In daily life, there are extremely minute amounts of chloroform in the tap water caused by water disinfectants. It has no effect on the human body due to the extremely low dose. So we couldn't detect the odor of chloroform from tap water.
Solvent
It was once a universal solvent, but now it is used less because of its toxicity and because it is on the list of vulnerable chemicals. Most of the time chloroform is used because it cannot be replaced by methylene chloride under certain circumstances.
IV, Chloroform uses
What is chloroform used for?
4.1 Organic solvent
It is a non-flammable organic solvent commonly used in laboratories and industry. The most important role of chloroform is as a solvent, can dissolve a variety of organic substances (including grease, wax, etc.), but also can dissolve organic glass, resin, paint, rubber, and other polymer materials, and can be used as their adhesives.
4.2 Organic synthesis
Chloroform is also an important raw material for organic synthesis, and is the raw material for the manufacture of difluorochloromethane (Freon-22) and polytetrafluoroethylene (PTFE), and can also be used as an extractant for penicillin, essential oils, alkaloids and so on.
The greatest use of chloroform is in the production of polytetrafluoroethylene (PTFE), a relatively heat-resistant plastic known for the production of non-stick pan coatings.
4.3 Extraction
Chloroform is very effective at extracting substances from plant material and is therefore useful in the pharmaceutical industry to extract drugs or drug precursors from plants.
The equipment and its sketch
4.4 Chemical Analysis
Chloroform is also used in chemical analysis to isolate compounds from samples and can be used in the man-made synthesis of many organic chemicals.
4.5 Deuterium Reagent (Chloroform d)
Chloroform deuterium (CDCl3) is also one of the most common deuterium reagents, is inexpensive, and is widely used in nuclear magnetic resonance (NMR) spectroscopy.
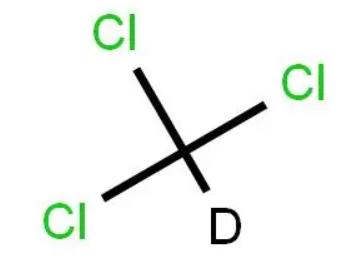
4.6 Chloracarbon
Chloroform is also a major source of chlorocarbon, which can be formed by the reaction of chloroform with an aqueous solution of NaOH in the presence of a phase transfer catalyst.
Dichlorocarbine can act on the o-formylation of aromatic rings (e.g. phenols) to form aromatic aldehydes in the Reimer-Tiemann reaction. Dichlorocarbine can also react with olefins to form cyclopropane derivatives. In the Kharasch reaction, chloroform in the presence of an olefin generates the CHCl2 radical.
4.7 Halogenation
As a reaction environment, it is commonly used for halogenation, since its own hydrogen has been heavily substituted.
4.8 Reduce Sulphuric Acid Carbonation
Chloroform is also used in a variety of reactions mixed with concentrated sulphuric acid, because of its high density and low melting point, and because of its miscible emulsion with sulphuric acid, which substantially reduces sulphuric acid carbonation.
V, Eight technical routes for the production of chloroform.
5.1 Made from acetaldehyde.
Lime water chlorination to get bleach solution, and then reacts with acetaldehyde solution to generate crude chloroform.
5.2 Use acetone as raw material and calcium hypochlorite reaction.
5.3 Methane as raw material, direct chlorination.
This reaction is a strong exothermic reaction. In order to increase the rate of chloroform and reduce the by-products (methylene chloride, methylene chloride, and carbon tetrachloride), people should strictly control the molar ratio of methane and chlorine and temperature.
5.4 Hexachloroacetone reacts with sodium hydroxide to make chloroform.
CCl3COCCl3+NaOH→ Chloroform+CCl3COONa
5.5 Chloroform can be made from carbon tetrachloride by using iron and water at high temperatures.
5.6 Carbon tetrachloride as raw material, under ultraviolet light irradiation, and ethanol reaction, can be made chloroform.
CCl4 + CH3CH2OH → Chloroform + ClCH2CH2OH
5.7 Carbon tetrachloride is catalyzed by zinc powder, with hydrogen reduction.
5.8 Trichloroacetaldehyde method, it is, trichloroacetaldehyde reacts with calcium hydroxide to form Chloroform.
Back to Top


